Appendicitis Disease Overview:
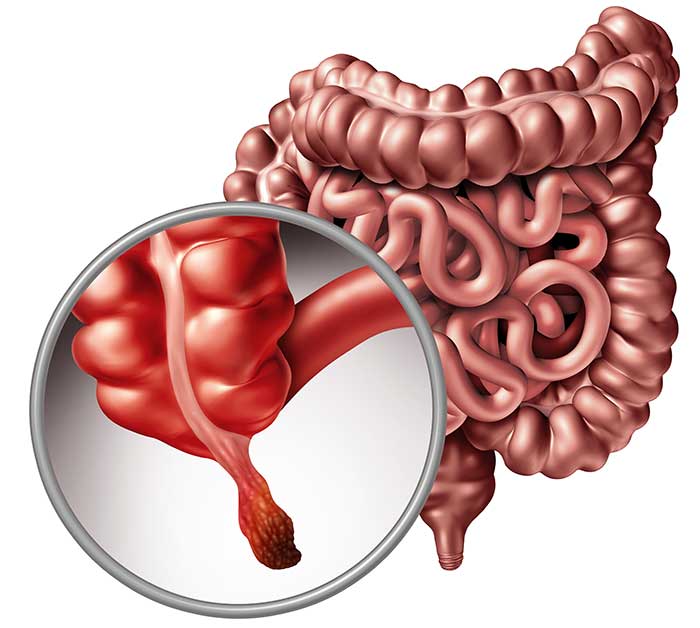
Appendicitis Disease is a medical condition characterized by the inflammation of the appendix, a small organ located in the lower right abdomen. This condition demands prompt medical attention, as a delayed diagnosis and treatment can lead to severe complications, including the rupture of the appendix. In this article, we will delve into various aspects of appendicitis, covering diagnostic analysis, treatment options, regulatory frameworks, competitive landscape, market trends, and clinical trial data assessment.
The Market Competitors Listed Below are Revolutionizing Healthcare with Innovative Inventions:
Diagnostic Market Players:
Blood Tests
· Sun Pharmaceuticals Industries Ltd
· Cipla Limited
· Dr. Reddy’s Laboratory
· Max Healthcare
· Fortis Healthcare
· SRL Diagnostics
Imaging Tests
· DiagnoTech Corporation
· Siemens Healthineers
· Med Imaging Solutions
· Radiance Imaging System
· Hitachi Ltd.
· Esaote S.p.A.
Treatment Market Players:
· Shandong Octagon Chemicals Ltd.
· Reyoung Pharmaceuticals
· Sichuan Kelun Pharmaceutical Co Ltd.
· Cipla Ltd.
· Albert David
· Pfizer
· Johnson and Johnson
· Manus Aktteva Biopharma LLP
· Pfizer
· DEV LIFE CORPORATION PVT LTD
· Aden Healthcare
· Anant Pharmaceuticals Pvt. Ltd.
Appendicitis Diagnostic Analysis:
Timely and accurate diagnosis is crucial in managing appendicitis effectively. Physicians typically rely on a combination of clinical evaluation, medical history, and imaging studies. Common diagnostic tools include physical examinations, blood tests, and imaging techniques such as ultrasound and computed tomography (CT) scans. However, advancements in diagnostic technologies continue to refine the accuracy and efficiency of appendicitis diagnosis.
Appendicitis Treatment Analysis:
Once diagnosed, the primary mode of treatment for appendicitis is surgical removal of the inflamed appendix, known as an appendectomy. This can be performed through traditional open surgery or minimally invasive laparoscopic techniques. In some cases, when the appendix has already ruptured, the treatment may involve additional measures such as drainage of abscesses and administration of antibiotics.
Regulatory Framework for Appendicitis:
Regulatory bodies play a crucial role in ensuring the safety and efficacy of treatments for appendicitis. Approval processes and guidelines set by health authorities, such as the Food and Drug Administration (FDA) in the United States or the European Medicines Agency (EMA) in Europe, help establish standards for the development and marketing of appendicitis-related drugs and devices. Staying compliant with these regulations is essential for healthcare stakeholders.
https://www.diseaselandscape.com/gastrointestinal/appendicitis-disease-services-market-strategy
Competitive Analysis:
The landscape of appendicitis treatment is dynamic, with pharmaceutical companies and medical device manufacturers actively involved in research and development. Competition in the market includes the development of innovative surgical techniques, antibiotics, and imaging technologies. Key players are constantly striving to introduce advanced solutions that enhance diagnostic accuracy, minimize invasiveness, and improve patient outcomes.
Market Trends:
Several trends shape the appendicitis market, including a growing emphasis on minimally invasive surgical approaches, advancements in diagnostic imaging technologies, and the development of targeted pharmaceutical interventions. Additionally, a focus on patient-centric care and the integration of digital health solutions for remote monitoring and follow-up are gaining traction in the management of appendicitis.
Clinical Trial Data Assessment:
Ongoing clinical trials provide valuable insights into emerging treatments, technologies, and therapeutic approaches for appendicitis. Analyzing clinical trial data helps assess the safety and efficacy of experimental interventions, contributing to the overall understanding of the disease and guiding future treatment modalities.
Conclusion:
Appendicitis remains a common and potentially serious medical condition, necessitating a multidimensional approach for effective management. Advances in diagnostic techniques, surgical procedures, and pharmaceutical interventions continue to evolve, promising improved outcomes for patients. The collaboration between healthcare professionals, regulatory bodies, and industry stakeholders is pivotal in addressing the challenges associated with appendicitis and ensuring the development of safe and effective treatments in the years to come.
Browse Through More Gastrointestinal Disease Research Reports
Related Reports:
Unmasking the Enigma: Solid Tumors and Their Intricate Nature
Unravelling Hemophilia Disease: Understanding a Lifelong Challenge
Conquering Challenges: The Journey with Spinal Muscular Atrophy (SMA) Disease
Demystifying Demodex Blepharitis: Causes, Symptoms, and Treatment
Breathing Easier: A Comprehensive Guide to Cystic Fibrosis Disease